
When polystyrene foam is used, some paints may react negatively with it. The arrangement of protons and neutrons in an atom does not have a set pattern or order, and the protons and neutrons in an atom are constantly moving. Attach 10 red balls, or electrons, to the shape of a ball with bamboo skewers. As you can see, the nucleus of the atom contains ten protons and ten neutrons. A variety of materials can be used to build a model of the neon atom. Neon gas is commonly used as an advertising gas, as well as in lighting arrestors, high voltage indicators, and other commercial applications. In 1898, William Ramsay and Morris Travers discovered neon. With a little bit of patience and creativity, you can create a 3D neon atom model that looks stunning and helps you learn about the structure of atoms. In this guide, we’ll go over the materials you need, the steps to follow, and a few tips to make the process easier. With a few simple materials and a little bit of time, you can make a neon atom model that looks amazing and helps you learn about the structure of atoms. Not only is it a fun and creative project for kids, but it can also be used as a teaching tool in the classroom. It is a great way to learn about the structure of atoms, as well as the periodic table. On the other hand neon, the noble gas, immediately preceding it in the periodic table, requires 2081 kJ/mol or 21.56 eV/atom.Creating a 3D neon atom model is a fun and educational activity. Ionization energy increases across a row on the periodic maximum for the noble gases which have closed shells.įor example, sodium requires only 496 kJ/mol or 5.14 eV/atom to ionize it.Ionization energy is lowest for the alkali metals which have a single electron outside a closed shell.Therefore the electronegativity is greatest at the top-right of the periodic table and decreases toward the bottom-left. Elements with high ionization energies have high electronegativities due to the strong pull exerted by the positive nucleus on the negative electrons. Electrons with low ionization energies have low electronegativities because their nuclei do not exert a strong attractive force on electrons. Ionization energy is is related with electronegativity and electron affinity. In general, the ionization energy increases moving up a group and moving left to right across a period. Helps to understand reactivity of elements (especially metals, which lose electrons). For example, only 7.38 eV is required to remove the outermost electron from a lead atom, while 88,000 eV is required to remove the innermost electron. Some of these electrons are more tightly bound in the atom than others. 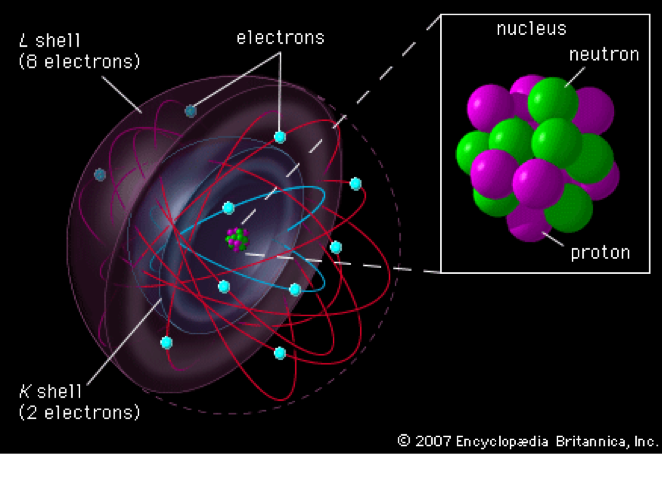
The electrons that circle the nucleus move in fairly well-defined orbits. There is an ionization energy for each successive electron removed. 
The nth ionization energy refers to the amount of energy required to remove an electron from the species with a charge of ( n-1). The ionization energy associated with removal of the first electron is most commonly used. Where X is any atom or molecule capable of being ionized, X + is that atom or molecule with an electron removed (positive ion), and e − is the removed electron.Ī Neon atom, for example, requires the following ionization energy to remove the outermost electron. Ionization energy, also called ionization potential, is the energy necessary to remove an electron from the neutral atom. First Ionization Energy of Neon is 21.5645 eV.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
